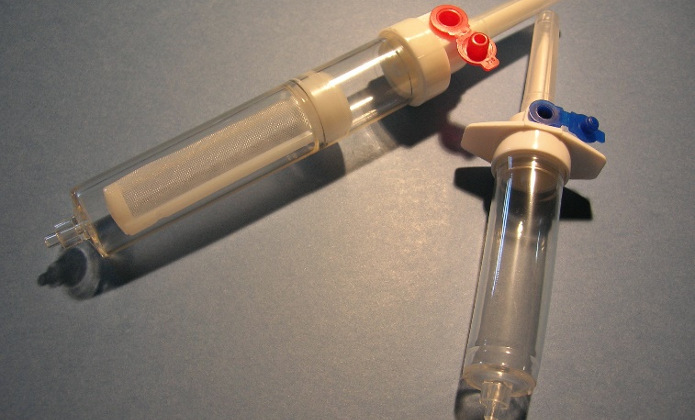
WHAT DO WE OFFER
A good knowledge of the market combined with an ability to identify the best components allow us to offer high-quality products at reasonable prices
DEVICES
Read More
ASSEMBLIES
Read More
COMPONENTS
Read More

WHAT WE STAND FOR
JCM MED is composed of a team of experimented professionals with one goal : to manufacture and market high quality sterile medical devices at reasonable prices. This is achieved through an ability to identify the best components and establish partnerships.
What is at stake is customer's satisfaction as much as a long-term social and economical project which is meant to establish the companies’ international renown in its sector.
OUR QUALITY POLICY
Our general policy takes into account market data as well as regulatory aspects
and our commitment is therefore to maintain and improve :
- The safety of our products
- The quality of our products and services
- The quality of our suppliers
- Our customer’s satisfaction
- Our international recognition

Our general policy takes into account market data as well as regulatory aspects and our commitment is therefore to :
- - Implement and maintain the efficiency of a quality management system that conforms to the EN ISO 13485 : 2016 standard.
- Make sure that our medical devices meet the requirements of the CE 93/42 Directive and maintain the surveillance of incidents and incident's risks that may result from the use of the sterile medical devices after their launch on the market.
Since our quality policy must be adapted to our organization, we regularly carry out reviews in order to verify its adequacy to our needs.
Our sterile medical devices are manufactured in different parts of the world. We regularly carry out audits of the Quality Management system and of the manufacturing premises, so that to ensure that the products are best manufactured in those locations. The QM System is certified against the ISO 13485 standard and all our sterile devices are CE marked. The products are manufactured in ISO-7 and ISO-8 class clean rooms.